 13Al, 14Si, 11Na, 12Mg, 16S Which of the above elements has the highest metallic character? 19K, 3li, 11Na, 4Be Which of these atoms has smallest atomic radius? 11Na, 15P, 17Cl, 14Si, 12Mg which of these has largest atoms?į. 4Be, 6C, 8O, 5B, 13Al Which is the most electropositive element among these?Īmong these, 13Al is the most electropositive element.Į. 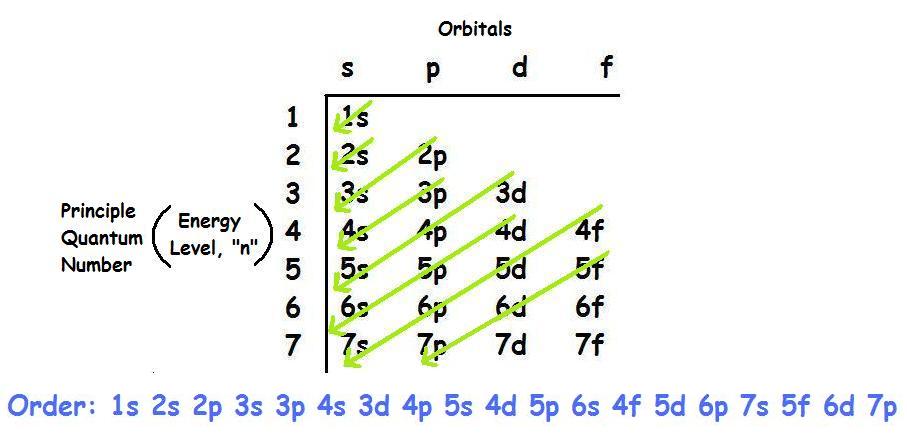
7N, 6C, 8O, 5B, 13Al Which is the most electronegative element among these?Īmong these, 8O is the most electronegative element.ĭ. Which of these elements belong to the second group?Įlements belongs to the 2nd group: 4Be and 20Ca.Ĭ. 3Li, 14Si, 2He, 11Na, 15P which of these elements belong to be period 3?Įlements belong to the 3rd period: 14Si, 11Na and 15P.ī. Answer the following question with explanation.Ī. Write down the electronic configuration of the following elements from the given atomic numbers. With which of the following elements would this element resemble? (Atomic numbers are given in the brackets)Ĭlass 10 Science 1 Chapter 2 Periodic Classification Of Elements Question 4. To which period does this element belong?ĭ. What is the atomic number of this element?Ĭ. (d) In which block of the modem periodic table are the nonmetals found?Ĭlass 10 Science Chapter 2 Periodic Classification Of Elements Notes Question 3.Īn element has its electron configuration as 2, 8, 2. which of the following elements be present in the same group as X. This compound is a solid having high melting point. (c) Molecular formula of the chloride of an element X is XCl. This means that their position in the modern periodic table is in……. (b) Alkaline earth metals have valency 2. (a) The number of electrons in the outermost shell of alkali metals is……. Periodic Classification Of Elements Class 10 Maharashtra Board Question 2.Ĭhoose the correct option and rewrite the statement: Lightest and negatively charged particle in all the atoms Properties of the eighth element similar to the first Sequential change in molecular formulaeĪverage of the first and the third atomic mass Properties of the eighth element similar to the firstį. Average of the first and the third atomic massĭ. Lightest and negatively charged particle in all the atomsĬ. Rearrange the columns 2 and 3 so as to match with the column 1. Periodic Classification Of Elements Question 1. Maharashtra State Board Class 10 Science Solutions Part 1 Chapter 2 Periodic Classification of Elements The electronic configuration of Cobalt will be 1s2 2s2 2p6 3s2 3p6 3d7 4s2.Balbharti Maharashtra State Board Class 10 Science Solutions Part 1 Chapter 2 Periodic Classification of Elements Notes, Textbook Exercise Important Questions and Answers. How do you write the electron configuration for Cobalt? The electronic configuration of Cobalt will be 1s2 2s2 2p6 3s2 3p6 3d7 4s2. What is the electronic configuration of Cobalt 27? What is the boiling Point of Cobalt in Kelvin?īoiling Point of Cobalt in Kelvin is 3200 K. Melting Point of Cobalt in Kelvin is 1768 K. What is the melting Point of Cobalt in Kelvin? What is the boiling Point of Cobalt?īoiling Point of Cobalt is 3200 K. Cobalt has 27 electrons out of which 4 valence electrons are present in the 3d7 4s2 outer orbitals of atom. 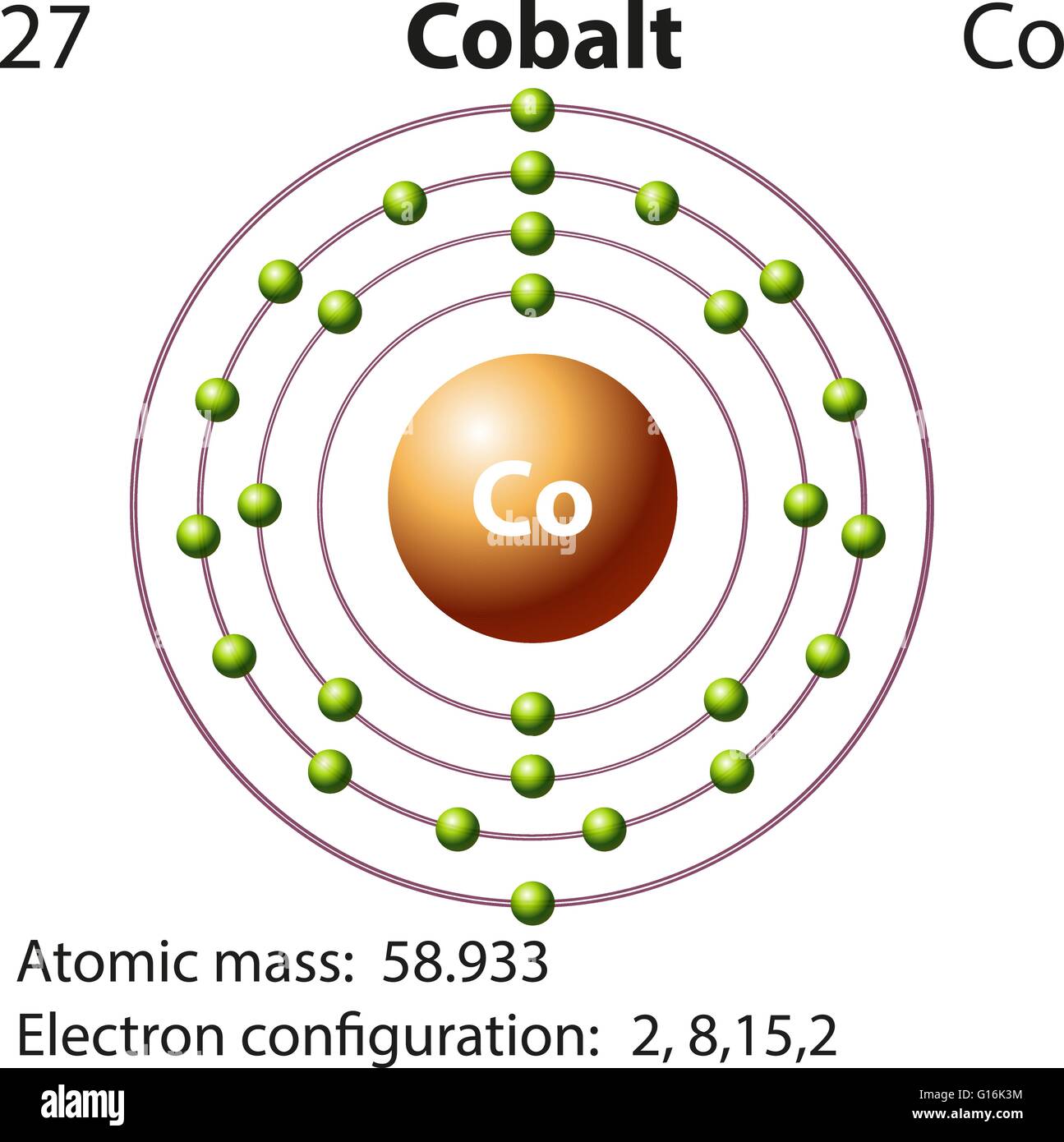
How many valence electrons does a Cobalt atom have?Ĭobalt has 4 valence electrons. What is the color of Cobalt?Ĭobalt is of Gray color. It is located in group 9 and period 4 in the modern periodic table. Cobalt is the 27 element on the periodic table. What is the position of Cobalt in the Periodic Table?Ĭobalt is a chemical element with the symbol Co and atomic number 27. Cobalt is a chemical element with symbol Co and atomic number 27. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Cobalt is 3d7 4s2. What is the abbreviated electronic configuration of Cobalt? The electronic configuration of Cobalt is 1s2 2s2 2p6 3s2 3p6 3d7 4s2. What is the electronic configuration of Cobalt? Optical Properties of Cobalt Refractive IndexĪcoustic Properties of Cobalt Speed of SoundĬobalt Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofCobalt Electrical ConductivityĬobalt Heat and Conduction Properties Thermal Conductivity Hardness of Cobalt - Tests to Measure of Hardness of Element Mohs HardnessĬobalt is Conductor of electricity. Refer to below table for Cobalt Physical Properties DensityĨ.9 g/cm3(when liquid at m.p density is $7.75 g/cm3) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
